
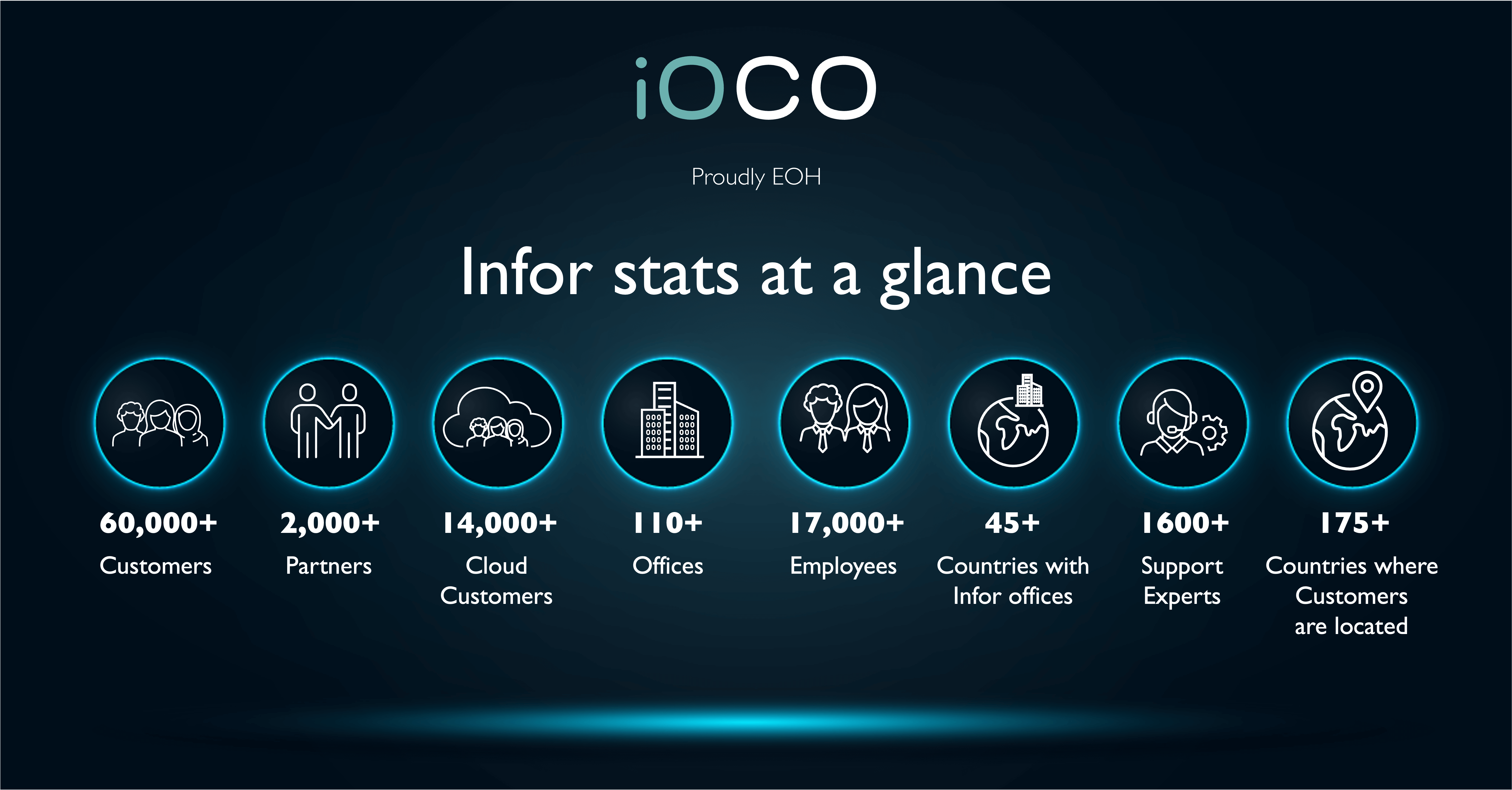
Infor offers a comprehensive and fully integrated stack of cloud applications, platform services, and engineered systems. iOCO Software Distribution is Infor’s largest and longest standing South African Partner who delivers Infor ERP and extended solutions to the local market.
Infor - the industry cloud company
ERP
SUPPLY CHAIN
WORKFORCE
MANAGEMENT
HCM
FINANCIALS
ASSET MANAGEMENT
Industry focus
VERTICAL EXPERTISE IS AT THE CORE OF OUR PRODUCTS, TECHNOLOGY, AND DEPLOYMENT CAPABILITIES
Innovation
OUR PRODUCTS AND SERVICES PROVIDE A PLATFORM TO TRANSFORM OPERATIONS AND ACCELERATE INNOVATION
Customer centricity
OUR CUSTOMER EXPERIENCE IS DEFINED BY LISTENING, TRANSPARENCY, AND DELIVERING ON OUR COMMITMENTS

Infor solutions are built for the cloud and reduce company overhead. Built to industry needs, CloudSuites are cost-effective and reduce implementation time and is the only multi-tenant cloud enterprise solution infrastructure that delivers industry, cross-industry, operational, and technology platform capabilities.
Infor CloudSuite™ software delivers an unmatched user experience, with ERP at its core—ready for the last mile, and ready to help SMBs and enterprise-level organizations take full advantage of the business benefits of cloud technology.
CloudSuite™ Automotive
CloudSuite Automotive provides a comprehensive suite of industry-proven automotive solutions, including a core ERP combined with high-value extension applications, to provide automotive suppliers the software they need to meet aggressive schedules, globalisation, cost challenges, and increasingly complex component production.
CloudSuite™ Distribution
Infor CloudSuite Distribution provides comprehensive, end-to-end functionality that helps boost performance across your entire organization. It's also continuously getting better, with new capabilities every 30 days. Just enable new features as they become available to take advantage of additional capabilities
CloudSuite™ Industrial
Discrete manufacturers around the world use Infor® Cloudsuite Industrial on-premises and in the cloud to simplify and automate their complex manufacturing processes. Offering a range of capabilities from advanced planning and materials management to financials, Cloudsuite Industrial ® delivers everything manufacturers need in a standard package.
CloudSuite™ Industrial Enterprise
A technology backbone for modernizing industrial enterprises Infor CloudSuite Industrial Enterprise is the technology backbone you need to modernise your business. Boost performance and efficiency in areas that are essential to your manufacturing organisation – improving customer engagements, your operations and the supply chain.
CloudSuite™ Healthcare
A complete operational platform designed for healthcare Infor CloudSuite Healthcare is a complete software platform designed for healthcare—that encompasses a comprehensive suite of business solutions unique to healthcare, including finance, planning, and supply chain. Additional capabilities include human capital and asset management.
CloudSuite™ Facilities
Facility managers, service providers, building owners, and tenants all have a vested interest in transparent operations, regulatory and SLA compliance, effective cost management, long-term asset value, and a workforce that creates quality service for tenants and customers.
Meet Infor CloudSuite™ Facilities Management, an award-winning, secure set of proven solutions that includes a core enterprise asset management system combined with high-value extension applications. Deployed in the cloud, it's designed for buildings of all types, from public, commercial, and industrial spaces to offices and multi-unit residential buildings.
Solutions that add value
iOCO Software Distribution takes pride in our people, because it is their industry knowledge and product expertise, which enable us to service our customers’ needs and build a long-lasting relationship as a digital journey partner. As an organisation we are able to channel this expertise and deliver value adding solutions.
News




Infor Agility – the difference
What is Infor Agility?
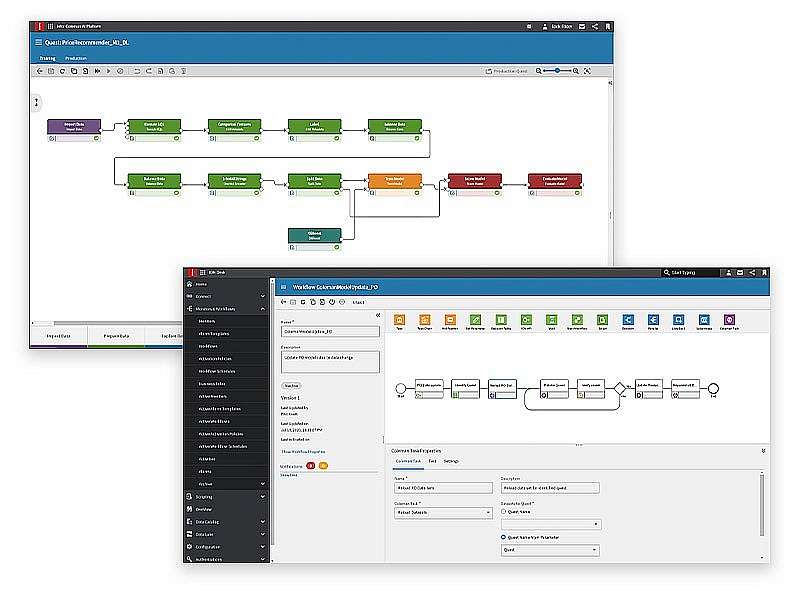
Infor® Agility is a program that combines aspects of Agile Methodology with advanced Implementation Accelerators (IA 4.0), Process Intelligence, Migration Factory, Testing as a Service (TaaS), and Consumerized Learning. This takes a prescriptive approach, instead of an “implement as you please” approach.
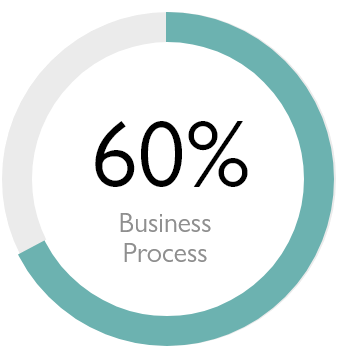


Infor Building Blocks
To drive constant innovation in the enterprise, Infor business applications are specialised by industry and built for the cloud to enable a global supply chain, networked analytics, and an artificial intelligence-led user experience.






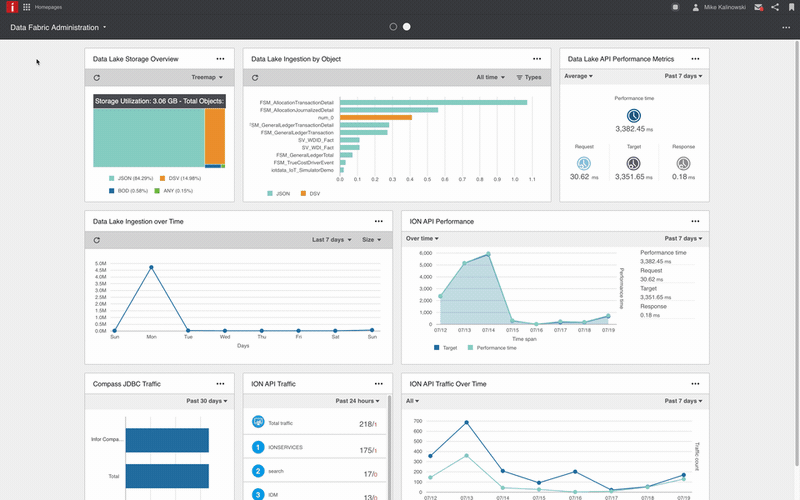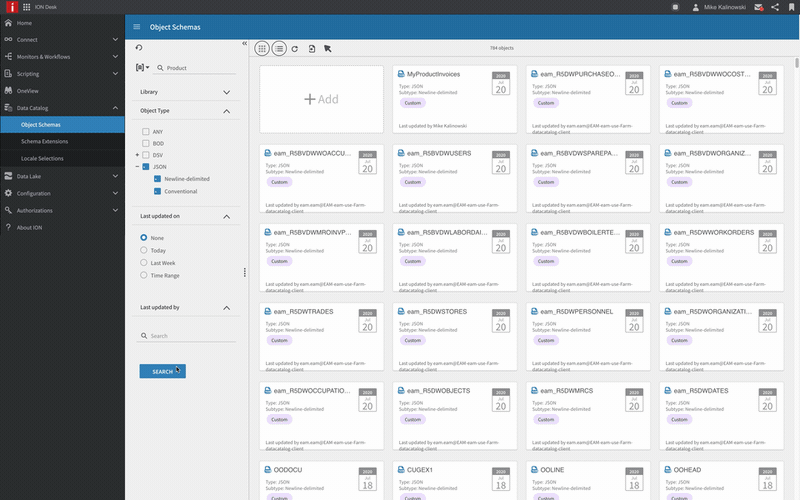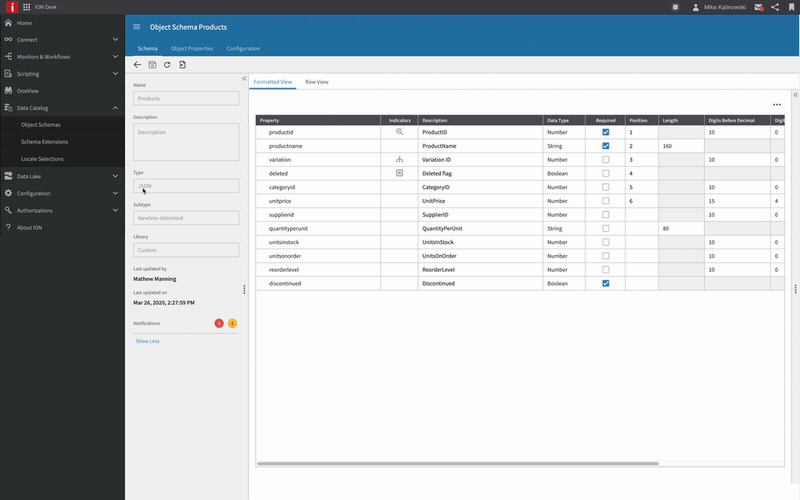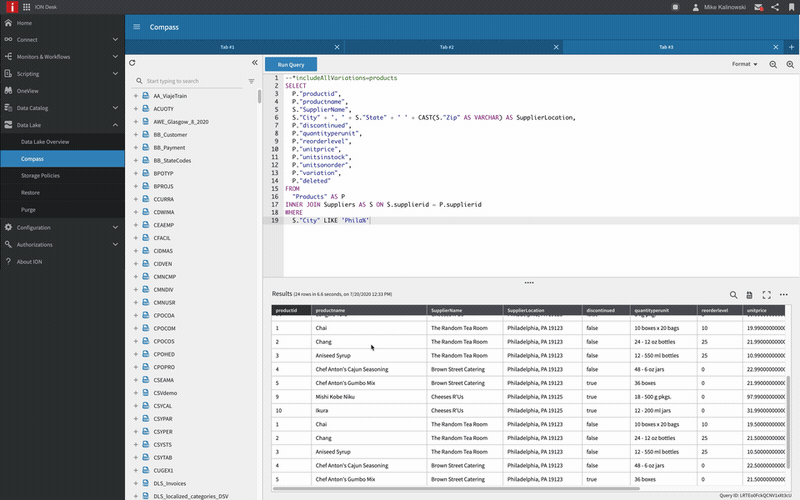
Infor OS
Infor® OS is a cloud operating platform that seamlessly connects services to provide a robust technology framework that serves as the foundation of innovation for the entire business ecosystem.
Unified infrastructure
Proven multi-domain enterprise technology platform providing the foundation for business excellence:
User-centered experience
Providing an end-to-end framework for creating competitive advantage and differentiation through intuitive UX
Future-enabled capabilities
Continuously built to adapt so that innovation is no longer a choice
Enterprise software solutions—built for the cloud, built for your industry.
Elite customer support
Client specific managed services
24/7 mobile access
Industry ERP solutions
With iOCO as your digital journey partner, we are building the future with you regardless of what industry you are in, Infor is ready to provide adaptable and comprehensive solutions that drive business results. Infor software applications are built on decades of experience in key industry verticals, by experts who understand the unique challenges that face your organization.
Enterprise Asset Management
Flexible and configurable to enterprise needs Infor® EAM digitizes maintenance with built-in industry-specific capabilities for manufacturing, transportation, public sector, healthcare, and more. Native mobile applications significantly improve field efficiency, providing your team with time to focus on more strategic initiatives.
Enterprise Performance Management
Budgeting, forecasting and planning – essential for everyday business management. Full business visibility. Gain a complete, real-time view of your business performance with Infor Dynamic Enterprise Performance Management® (Infor d/EPM®). Combining intelligent business tools and financial planning capabilities in an EPM software solution, d/EPM enables you to report with confidence, measure past and current performance, and forecast future activities.
Human Capital Management (HCM)
Infor® Human Resources is a powerful set of cloud-based human capital management (HCM) software solutions that empower your people to deliver streamlined processes with remarkable experiences. This solution replaces complex processes, workflows, and systems with sophisticated, yet intuitive technology. Give your HR professionals the ability to connect your business and talent objectives to create significant value across your organization.
Advanced Warehousing
When speed and accuracy are critical to customer satisfaction, businesses must rely on their warehouses to keep the supply chain moving. Infor WMS provides unprecedented visibility into inventory, orders, equipment, and people to empower organizations to enrich service levels and increase product velocity.
Infor WMS lets businesses manage distribution center activities holistically. The solution combines warehouse fulfillment with embedded labor management and 3D visual analysis to reduce complexity and support enhanced operational execution. Designed with configurability and intuitive use, Infor WMS is ready to power next-generation, technology-enhanced, global warehousing operations.
Trusted by:











Pick your platform, start the conversation
Ready to talk about how we can help you?
Infor products
Cloud Suites
- CloudSuite™ Aerospace & Defense
- CloudSuite™ Automotive
- Infor CPQ
- CloudSuite™ Corporate
- Infor CRM
- CloudSuite™ Distribution
- CloudSuite™ Distribution Enterprise
- CloudSuite™ Equipment
- Infor Expense Management
- CloudSuite™ Facilities Management
- CloudSuite™ Fashion
- CloudSuite™ Food & Beverage
- CloudSuite™ Healthcare
- CloudSuite™ Industrial Enterprise
- CloudSuite™ Public Sector
- CloudSuite™ Retail
- Infor WMS
- Coleman AI
- Cloverleaf Integration Suite
- Complete Billing System
- Customer Experience Suite
- Data Lake
- Infor Demand Management
- Infor Distribution SX.e
- Infor Dynamic Enterprise Performance Management (Infor d/EPM)
- Hospitality Management Solution (HMS)
- Infor Hospitality Price Optimizer
- HR Benefits
- HR Case Management

